The Flaviviradae is a large family of viral pathogens responsible for causing severe disease and mortality in humans and animals. The family consists of three genera: Flavivirus, Pestivirus and Hepacivirus.
The Flavivirus genus, which is the largest of the three, contains more than 70 viruses including Dengue Virus (DV), Japanese Encephalitis Virus (JEV), West Nile Virus (WNV), Yellow Fever Virus (YFV) and Zika Virus (ZIKV).
Flaviviruses show morphological uniformity with an icosahedral capsid and closefitting, spiked envelope. The size of the capsid is about 30 nm and the whole virion measures 45 nm. The genome of the flaviviruses is a singlestranded sense RNA about 10 kb in size.
It codes for 3 structural proteins: capsid (C protein), membrane (M, which is expressed as prM, the precursor to M and envelope (E protein) and 7 nonstructural proteins: NS1, NS2A, NS2B, NS3, NS4A, NS4B and NS5 (Figure 1 a.b).
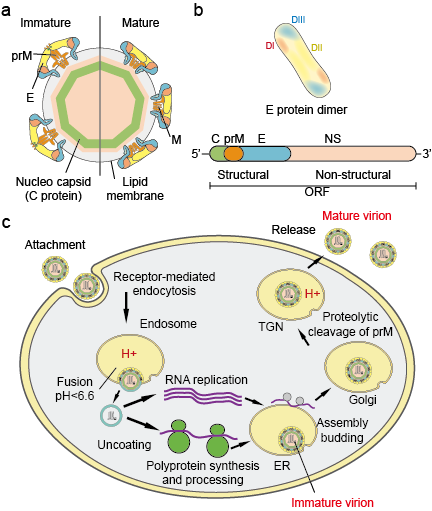
Figure 1. Flavivirus particles (a), proteins (b) and life cycle (c).
Browse All Flavivirus Related Products
Flavivirus life cycle: Virions attach to the surface of a host cell and subsequently enter the cell by receptor-mediated endocytosis (Figure 1c). Several primary receptors and low-affinity co-receptors for flaviviruses have been identified.
Acidification of the endosomal vesicle triggers conformational changes in the virion, fusion of the viral and cell membranes, and particle disassembly. Once the genome is released into the cytoplasm, the positive-sense RNA is translated into a single polyprotein that is processed co- and post-translationally by viral and host proteases.
Genome replication occurs on intracellular membranes. Virus assembly occurs on the surface of the endoplasmic reticulum (ER) when the structural proteins and newly synthesized RNA buds into the lumen of the ER. The resultant non-infectious, immature viral and subviral particles are transported through the trans-Golgi network (TGN).
The immature virion particles are cleaved by the host protease furin, resulting in mature, infectious particles. Subviral particles are also cleaved by furin. Mature virions and subviral particles are subsequently released by exocytosis.
Flavivirus Epidemiology: Mosquito-borne flaviviruses are transmitted in nature in one or more distinct or overlapping cycles that include a mosquito vector, generally Aedes mosquitoes for YFV and DENV and Culex mosquitoes for JEV and WNV, and a mammalian or avian host.
Transmission between mosquitoes and vertebrate hosts is termed horizontal transmissions and causes disease in vertebrates. In contrast to horizontal transmission, mosquito-borne flaviviruses can be maintained in the environment through vertical, i.e.
, transgenerational, transmissions which allow the spread of flaviviruses solely in mosquitoes. The most direct evidence supporting the vertical transmission of mosquito-borne flaviviruses is derived from the isolation of virus from infected larvae presumably through transovarial transmission.
This observation is consistent with the detection of viral antigens in ovarian tissues of infected mosquitoes. (Figure 2)

Figure 2. Three typical mosquitos that transmit disease.
Flaviviruses have a global distribution, and some members of the genus constitute a major public health issue (e.g.
, yellow fever virus [YFV], dengue virus [DENV], West Nile virus [WNV] and Japanese encephalitis virus [JEV]), with high morbidity and/or mortality.
In the last decade, flaviviruses have demonstrated an increased prevalence, posing a risk for more than 3 billion people worldwide, which makes them a paradigm of emerging diseases.
In the last 50 years many flaviviruses, such as dengue, West Nile, and yellow fever viruses, have exhibited dramatic increases in incidence, disease severity and/or geographic range. Environmentally derived viral pathogens display relatively uniform epidemiologic characteristics.
Mosquitoes, ticks, and biting flies serve as the vectors for most human viral diseases.
Human disease occurs when vectors are active, typically in spring, summer, and fall in temperate climates, and often displays distinct epidemiological characteristics that correspond to the habitat of the vector (Figure 3).

Figure 3. The epidemic area of 5 typical flavivirus.
Manifestations: Flaviviruses vary widely in their pathogenic potential and mechanisms for producing human disease (Table 1). Human infection with both mosquito-borne and tick-borne flaviviruses is initiated by deposition of virus through the skin via the saliva of an infected arthropod.
Virus replicates locally and in regional lymph nodes and results in viremia.
Major syndromes and examples of causative flaviviruses include: encephalitis (Japanese encephalitis), febrile illness with rash (dengue virus), hemorrhagic fever (Kyasanur Forest disease virus and sometimes dengue virus), and hemorrhagic fever with hepatitis (yellow fever virus).
Table 1 Overview of the Most Important Flaviviruses
| Yellow fever | Mosquito (Aedes) | See Figure 3a | Hemorrhagic fever |
| Dengue | Mosquito (Aedes, Stegomyia) | See Figure 3b | Dengue syndrome, DHF, DSS |
| West Nile fever | Mosquito (Culex), ticks (Argasidae) | See Figure 3c | Dengue syndrome, encephalitis |
| Japanese encephalitis | Mosquito (Culex) | See Figure 3d | Encephalitis |
| Zika | Mosquito (Aedes) | See Figure 3e | Microcephaly |
Diagnostics:
The Multiples Fates of the Flavivirus RNA Genome During Pathogenesis
Infections with flaviviruses constitute a major public health concern worldwide since they cause several human diseases with a wide range of symptoms that can potentially lead to lifelong impairment or even death.
The genus Flavivirus within the Flaviviridae virus family comprises almost 70 reported species including the most studied yellow fever virus (YFV), dengue virus (DENV), Zika virus (ZIKV), West Nile virus (WNV), Japanese encephalitis virus (JEV), and tick-borne encephalitis virus (TBEV).
The vast majority of flaviviral infections in humans occur through the biting by arthropods such as Aedes-type mosquitoes (mostly Aedes aegypti and Aedes albopictus) in the case of YFV, DENV, and ZIKV or Culex pipiens mosquitoes in the case of WNV. Vaccines do exist for YFV, DENV and TBEV.
However, in the case of DENV, the cause of the most prevalent arthropod-borne viral disease, the only available vaccine shows limited efficacy against all DENV serotypes and safety concerns have recently arisen in the Philippines in vaccinated children (Dyer, 2017).
Importantly, no antivirals against flaviviruses are currently available partly because of our limited understanding of their life cycle and pathogenesis when compared to other virus groups. Interestingly, it appears that the general features of the life cycle are conserved across flaviviruses.
Hence, there have been tremendous efforts by both industry and academia to identify or engineer antiviral drugs with a panflaviviral spectrum. This illustrates the importance of deciphering the molecular mechanisms underlying the flavivirus life cycle in order to identify novel antiviral targets.
The flavivirus life cycle is completely dependent on the cytoplasmic fate of only one RNA species, namely the genomic viral RNA (vRNA) whose replication entirely occurs in the cytoplasm and does not generate any DNA intermediates.
Most notably, vRNA contains all the genetic information necessary for optimal virus replication. Hence, targeting vRNA or viral processes involved in its metabolism constitutes an attractive strategy for the development of novel antivirals.
Moreover, fundamental virology often provides crucial insight into cellular machinery and processes at the molecular level. In this respect, flavivirus vRNA constitutes an exciting and excellent model for investigating the spatiotemporal regulation of RNA metabolism.
With that in mind, we focus this review on our current understanding of the multiple fates of vRNA and how it orchestrates the viral life cycle and creates a cellular environment favorable to infection.
Flaviviruses are enveloped positive-strand RNA viruses that presumably contain a single copy of the genome RNA. Following receptor-mediated endocytosis of the virion and fusion with the endosomal membrane (reviewed in Perera-Lecoin et al., 2013), the vRNA is uncoated and released into the cytosol.
The flaviviral vRNA genome contains all the genetic information required for efficient viral replication by hijacking the intracellular resources. With a single open reading frame, vRNA encodes an endoplasmic reticulum (ER)-associated transmembrane polyprotein (Figure 1A) (Garcia-Blanco et al.
, 2016; Neufeldt et al., 2018).

FIGURE 1. Schematic representation of flavivirus vRNA. (A) vRNA is composed of a 5′UTR, one single open reading frame and a 3′UTR. The position of the sequences encoding for the viral proteins within the polyprotein is indicated.
The bottom part of the figure shows in details the secondary structures of 5′UTR, capsid-coding region and 3′UTR. The different regions engaged in local pseudoknots and long-range RNA–RNA interactions are indicated and described in detail in the text. (B) Predicted structure of vRNA in its circularized conformation.
The coding sequence (except 5′ capsid coding region) is depicted with a dashed line.
Upon translation, the polyprotein is subsequently processed by both cellular and viral proteases to generate 10 mature viral proteins. Structural proteins Capsid (C), Envelop (E) and prM assemble new viral particles while non-structural (NS) proteins NS1, NS2A, NS2B, NS3, NS4A, NS4B, and NS5 are responsible for vRNA replication (Figure 1A).
vRNA synthesis relies on NS5, the RNA-dependent RNA polymerase as well as on critical vRNA secondary and tertiary structures. NS5 is also responsible for the capping of the neosynthetized vRNA. NS3 is a protease which, together with its co-factor NS2B, participates to the processing of the viral polyprotein.
It also possesses helicase, NTPase and triphosphatase activities, all required for efficient vRNA synthesis and capping. vRNA is then encapsidated into assembling viral particles which bud into the ER.
Assembled viruses egress through the secretory pathway where they undergo furin-mediated maturation in the Golgi apparatus, allowing fully infectious virions to be released via exocytosis (Apte-Sengupta et al., 2014; Neufeldt et al., 2018).
Flavivirus
Genus of virusesFlavivirusA TEM micrograph of Yellow fever virusZika virus capsid model, colored by chains, PDB entry 5ire[2]Virus classification (unranked):VirusRealm:RiboviriaKingdom:OrthornaviraePhylum:KitrinoviricotaClass:FlasuviricetesOrder:AmarilloviralesFamily:FlaviviridaeGenus:FlavivirusType speciesYellow fever virus[1]Species[1]
- Apoi virus
- Aroa virus
- Bamaga virus
- Bagaza virus
- Banzi virus
- Bouboui virus
- Bukalasa bat virus
- Cacipacore virus
- Carey Island virus
- Cowbone Ridge virus
- Dakar bat virus
- Dengue virus
- Edge Hill virus
- Entebbe bat virus
- Gadgets Gully virus
- Ilheus virus
- Israel turkey meningoencephalomyelitis virus
- Japanese encephalitis virus
- Jugra virus
- Jutiapa virus
- Kadam virus
- Kedougou virus
- Kokobera virus
- Koutango virus
- Kyasanur Forest disease virus
- Langat virus
- Louping ill virus
- Meaban virus
- Modoc virus
- Montana myotis leukoencephalitis virus
- Murray Valley encephalitis virus
- Ntaya virus
- Omsk hemorrhagic fever virus
- Phnom Penh bat virus
- Powassan virus
- Rio Bravo virus
- Royal Farm virus
- Saboya virus
- Saint Louis encephalitis virus
- Sal Vieja virus
- San Perlita virus
- Saumarez Reef virus
- Sepik virus
- Tembusu virus
- Tick-borne encephalitis virus
- Tyuleniy virus
- Uganda S virus
- Usutu virus
- Wesselsbron virus
- West Nile virus
- Yaounde virus
- Yellow fever virus
- Yokose virus
- Zika virus
Flavivirus is a genus of viruses in the family Flaviviridae. This genus includes the West Nile virus, dengue virus, tick-borne encephalitis virus, yellow fever virus, Zika virus and several other viruses which may cause encephalitis,[3] as well as insect-specific flaviviruses (ISFs) such as cell fusing agent virus (CFAV), Palm Creek virus (PCV), and Parramatta River virus (PaRV).[4] While dual-host flaviviruses can infect vertebrates as well as arthropods, insect-specific flaviviruses are restricted to their competent arthropods.[5]
Flaviviruses are named from the yellow fever virus, the type virus for the family; the word flavus means ‚yellow‘ in Latin and the yellow fever in turn is named from its propensity to cause yellow jaundice in victims.[6]
Flaviviruses share several common aspects: common size (40–65 nm), symmetry (enveloped, icosahedral nucleocapsid), nucleic acid (positive-sense, single-stranded RNA around 10,000–11,000 bases), and appearance in the electron microscope.
Most of these viruses are primarily transmitted by the bite from an infected arthropod (mosquito or tick), and hence are classified as arboviruses.
Human infections with most of these arboviruses are incidental, as humans are unable to replicate the virus to high enough titers to reinfect the arthropods needed to continue the virus lifecycle – humans are then a dead end host. The exceptions to this are the yellow fever, dengue, and zika viruses.
These three viruses still require mosquito vectors, but are well-enough adapted to humans as to not necessarily depend upon animal hosts (although they continue to have important animal transmission routes, as well).
Other virus transmission routes for arboviruses include handling infected animal carcasses, blood transfusion, sex, child birth and consumption of unpasteurised milk products.
Transmission from nonhuman vertebrates to humans without an intermediate vector arthropod however mostly occurs with low probability.
For example, early tests with yellow fever showed that the disease is not contagious.
The known non-arboviruses of the flavivirus family reproduce in either arthropods or vertebrates, but not both, with one odd member of the genus affecting a nematode.[7]
Structure
Viruses in Flavivirus are enveloped, with icosahedral and spherical geometries. The diameter is around 50 nm. Genomes are linear positive-sense RNA and non-segmented, around 10–11kb in length.[8]
| Flavivirus |
Flavivirus
Flavivirus Научная классификация
| Домен: | Вирусы[1] |
| Реалм: | Riboviria |
| Царство: | Orthornavirae |
| Тип: | Kitrinoviricota |
| Класс: | Flasuviricetes |
| Порядок: | Amarillovirales |
| Семейство: | Flaviviridae |
| Род: | Flavivirus |
Международное научное название
Flavivirus
Группа по Балтимору
IV: (+)оцРНК-вирусы
| Систематикана Викивидах | Изображенияна Викискладе |
|
Flavivirus (лат.) — род арбовирусов из семейства Flaviviridae. Типовой вид — вирус жёлтой лихорадки. Преимущественно циркулируют между членистоногими (клещи, комары) и млекопитающими (приматы, летучие мыши, грызуны, скот).
При укусе заражённого комара или клеща могут передаваться человеку, вызывая заболевания различной тяжести, варьирующей от бессимптомного протекания до угрожающих жизни геморрагических лихорадок и энцефалитов (лихорадка денге, лихорадка Западного Нила, лихорадка Зика, клещевой энцефалит, японский энцефалит, энцефалит Сент-Луис и др.).
При исследовании флавивирусов был обнаружен феномен антителозависимого усиления инфекции[2].
Классификация
На март 2020 года в род включают 53 вида[3]:
- Apoi virus
- Aroa virus
- Bagaza virus
- Banzi virus
- Bouboui virus
- Bukalasa bat virus
- Cacipacore virus
- Carey Island virus
- Cowbone Ridge virus
- Dakar bat virus
- Dengue virus
- Edge Hill virus
- Entebbe bat virus
- Gadgets Gully virus
- Ilheus virus
- Israel turkey meningoencephalomyelitis virus
- Japanese encephalitis virus
- Jugra virus
- Jutiapa virus
- Kadam virus
- Kedougou virus
- Kokobera virus
- Koutango virus
- Kyasanur Forest disease virus
- Langat virus
- Louping ill virus
- Meaban virus
- Modoc virus
- Montana myotis leukoencephalitis virus
- Murray Valley encephalitis virus
- Ntaya virus
- Omsk hemorrhagic fever virus
- Phnom Penh bat virus
- Powassan virus
- Rio Bravo virus
- Royal Farm virus
- Saboya virus
- Sal Vieja virus
- San Perlita virus
- Saumarez Reef virus
- Sepik virus
- St. Louis encephalitis virus
- Tembusu virus
- Tick-borne encephalitis virus
- Tyuleniy virus
- Uganda S virus
- Usutu virus
- Wesselsbron virus
- West Nile virus
- Yaounde virus
- Yellow fever virus
- Yokose virus
- Zika virus
Представители
Флавивирусы, переносимые комарами
Распространены преимущественно в тропических регионах:
- Вирус жёлтой лихорадки
- Вирус денге
- Вирус Западного Нила
- Вирус Зика
- Вирус японского энцефалита
Флавивирусы, переносимые клещами
Распространены в основном в регионах умеренного климата:
- Вирус клещевого энцефалита
- Вирус омской геморрагической лихорадки
- Вирус Повассан
Флавивирусы с неизвестным переносчиком
Выделены из различных млекопитающих, но не из членистоногих, цикл циркуляции неизвестен:
- Вирус Модок
Примечания
- ↑ Таксономия вирусов (англ.) на сайте Международного комитета по таксономии вирусов (ICTV).
- ↑ Hawkes R.A.
Enhancement of the infectivity of arboviruses by specific antisera produced in domestic fowls // Aust. J. Exp. Biol. Med. Sci. 1964. V. 43. P. 465—482.
- ↑ Таксономия вирусов (англ.
) на сайте Международного комитета по таксономии вирусов (ICTV). (Проверено 26 апреля 2020).
Литература
- Pierson, Theodore C. Flaviviruses : Chapter 26 // Fields Virology : [англ.] : in 2 vol. / Theodore C. Pierson, Michael S. Diamond. — 6th Ed. — Philadelphia : Lippincott Williams & Wilkins, 2013. — Vol. 1. — P. 747—794. — 2456 p. — ISBN 978-1-4511-0563-6.
- Lindenbach, Brett D. Flaviviridae : Chapter 25 // Fields Virology : [англ.] : in 2 vol. / Brett D. Lindenbach, Catherine L. Murray, Heinz-Jürgen Thiel … [et al.]. — 6th Ed. — Philadelphia : Lippincott Williams & Wilkins, 2013. — Vol. 1. — P. 712—746. — 2456 p. — ISBN 978-1-4511-0563-6.
| Это заготовка статьи по вирусологии. Вы можете помочь проекту, дополнив её. |
Источник — https://ru.wikipedia.org/w/index.php?title=Flavivirus&oldid=106851572
A recombinant platform for flavivirus vaccines and diagnostics using chimeras of a new insect-specific virus
Page 2
11 December 2019 Vol 11, Issue 522
-
- By Jody Hobson-Peters, Jessica J. Harrison, Daniel Watterson, Jessamine E. Hazlewood, Laura J. Vet, Natalee D. Newton, David Warrilow, Agathe M. G. Colmant, Carmel Taylor, Bixing Huang, Thisun B. H. Piyasena, Weng Kong Chow, Yin Xiang Setoh, Bing Tang, Eri Nakayama, Kexin Yan, Alberto A. Amarilla, Sarah Wheatley, Peter R. Moore, Mitchell Finger, Nina Kurucz, Naphak Modhiran, Paul R. Young, Alexander A. Khromykh, Helle Bielefeldt-Ohmann, Andreas Suhrbier, Roy A. Hall
- By Jörg Klufa, Thomas Bauer, Buck Hanson, Craig Herbold, Philipp Starkl, Beate Lichtenberger, Dagmar Srutkova, Daniel Schulz, Igor Vujic, Thomas Mohr, Klemens Rappersberger, Bernd Bodenmiller, Hana Kozakova, Sylvia Knapp, Alexander Loy, Maria Sibilia
- By Boris D. Heifets, Juliana S. Salgado, Madison D. Taylor, Paul Hoerbelt, Daniel F. Cardozo Pinto, Elizabeth E. Steinberg, Jessica J. Walsh, Ji Y. Sze, Robert C. Malenka
- By Xue Han, Matthew D. Vesely, Wendy Yang, Miguel F. Sanmamed, Ti Badri, Jude Alawa, Francesc López-Giráldez, Patricia Gaule, Sang Won Lee, Jian-Ping Zhang, Xinxin Nie, Ala Nassar, Agedi Boto, Dallas B. Flies, Linghua Zheng, Tae Kon Kim, Gilbert W. Moeckel, Jennifer M. McNiff, Lieping Chen
- By Amarnath Satheesh Marudamuthu, Yashodhar Prabhakar Bhandary, Liang Fan, Vijay Radhakrishnan, BreAnne MacKenzie, Esther Maier, Shwetha Kumari Shetty, M.R. Nagaraja, Venkadesaperumal Gopu, Nivedita Tiwari, Yajie Zhang, Alan B. Watts, Robert O. Williams III, Gerald J Criner, Sudhir Bolla, Nathaniel Marchetti, Steven Idell, Sreerama Shetty
- By Robert H. Lee, Raymond Piatt, Ankita Dhenge, María L. Lozano, Verónica Palma-Barqueros, José Rivera, Wolfgang Bergmeier
Page 3
Page 4
11 December 2019 Vol 11, Issue 522
-
- By Jody Hobson-Peters, Jessica J. Harrison, Daniel Watterson, Jessamine E. Hazlewood, Laura J. Vet, Natalee D. Newton, David Warrilow, Agathe M. G. Colmant, Carmel Taylor, Bixing Huang, Thisun B. H. Piyasena, Weng Kong Chow, Yin Xiang Setoh, Bing Tang, Eri Nakayama, Kexin Yan, Alberto A. Amarilla, Sarah Wheatley, Peter R. Moore, Mitchell Finger, Nina Kurucz, Naphak Modhiran, Paul R. Young, Alexander A. Khromykh, Helle Bielefeldt-Ohmann, Andreas Suhrbier, Roy A. Hall
- By Jörg Klufa, Thomas Bauer, Buck Hanson, Craig Herbold, Philipp Starkl, Beate Lichtenberger, Dagmar Srutkova, Daniel Schulz, Igor Vujic, Thomas Mohr, Klemens Rappersberger, Bernd Bodenmiller, Hana Kozakova, Sylvia Knapp, Alexander Loy, Maria Sibilia
- By Boris D. Heifets, Juliana S. Salgado, Madison D. Taylor, Paul Hoerbelt, Daniel F. Cardozo Pinto, Elizabeth E. Steinberg, Jessica J. Walsh, Ji Y. Sze, Robert C. Malenka
- By Xue Han, Matthew D. Vesely, Wendy Yang, Miguel F. Sanmamed, Ti Badri, Jude Alawa, Francesc López-Giráldez, Patricia Gaule, Sang Won Lee, Jian-Ping Zhang, Xinxin Nie, Ala Nassar, Agedi Boto, Dallas B. Flies, Linghua Zheng, Tae Kon Kim, Gilbert W. Moeckel, Jennifer M. McNiff, Lieping Chen
- By Amarnath Satheesh Marudamuthu, Yashodhar Prabhakar Bhandary, Liang Fan, Vijay Radhakrishnan, BreAnne MacKenzie, Esther Maier, Shwetha Kumari Shetty, M.R. Nagaraja, Venkadesaperumal Gopu, Nivedita Tiwari, Yajie Zhang, Alan B. Watts, Robert O. Williams III, Gerald J Criner, Sudhir Bolla, Nathaniel Marchetti, Steven Idell, Sreerama Shetty
- By Robert H. Lee, Raymond Piatt, Ankita Dhenge, María L. Lozano, Verónica Palma-Barqueros, José Rivera, Wolfgang Bergmeier